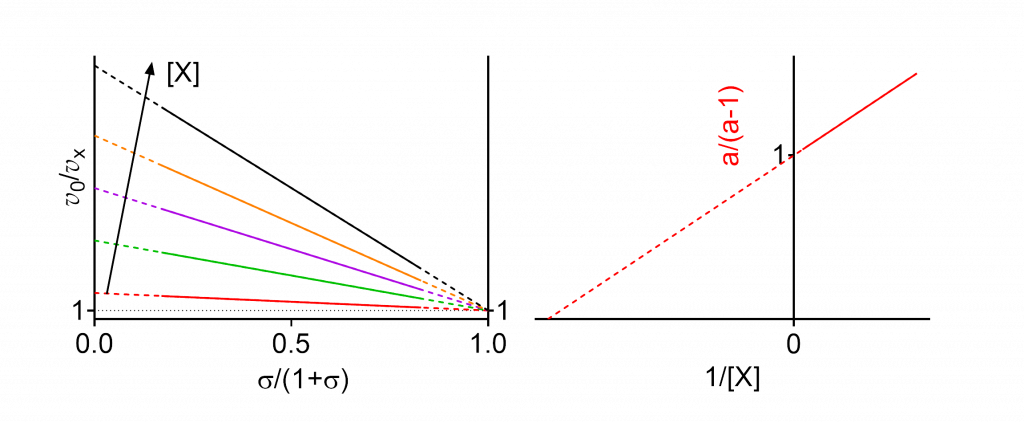
Linear specific inhibition

Featured examples
| # | Enzyme Species | EC no. | Modifier | Substrate(1) | Name given by authors(2) | Reference (3) |
|---|---|---|---|---|---|---|
| 1 | Alpha,alpha trehalase Chironomus riparius | 3.2.1.28 | 7-deoxy-epi-casuarine (Compound 6) | Trehalose | Pure competitive inhibition KX = 118 nM | D'Adamio (2015) |
| 2 | Coagulation factor IXa Homo sapiens | 3.4.21.27 | Aprotinin | L-Pyroglutamyl-L-prolyl-L-arginine-p-nitroanilide | Competitive inhibition KX = 1.23 μM | Ogawa (2005) |
| 3 | Cholinesterase (4) Homo sapiens | 3.1.1.8 | Tosyllysine chloromethyl ketone | Butyrylthiocholine | Pure competitive inhibition KX = 8 μM | Çokuğraş (2004) |
| 4 | Aldehyde oxidase Homo sapiens | 1.2.3.1 | 6-Chloroquinazolinone | Phtalazine | Strictly competitive inhibition KX = 5.4 μM | Barr (2011) |
| 5 | Spermine synthase Bos taurus | 2.5.1.22 | Spermine (reaction product) | Decarboxylated adenosylmethionine | Competitive inhibition KX ≈ 0.028 μM (5) | Pajula (1983) (6) |
| 6 | Tyrosinase (7) Homo sapiens | 1.14.18.1 | HTS05357 | L-Dopa | Purely competitive inhibition KX = 24.2 μM | Sun (2014) (8) |
| 7 | Dipeptidyl-peptidase IV Homo sapiens | 3.4.14.5 | Trp2,Ile3-Tat(1–9) MWIVDPNIE | Ala-Pro-p-nitroanilide | Competitive inhibition KX = 43.6 μM | Lorey (2003) (9) |
| 8 | Dipeptidyl-peptidase IV Homo sapiens | 3.4.14.5 | TXA2-R(1–9) MWPNGSSLG | Ala-Pro-p-nitroanilide | Competitive inhibition KX = 5.02 μM | Lorey (2003) (9) |
| 9 | Dipeptidyl-peptidase IV Homo sapiens | 3.4.14.5 | Met-Trp1-IL-2(1–8) MWPTSSSTK | Ala-Pro-p-nitroanilide | Competitive inhibition KX = 15.9 μM | Lorey (2003) (9) |
| 10 | Dipeptidyl-peptidase IV Homo sapiens | 3.4.14.5 | Met-Trp-Pro MWP | Ala-Pro-p-nitroanilide | Competitive inhibition KX = 24.5 μM | Lorey (2003) (9) |
| 11 | Acetylcholinesterase Torpedo californica | 3.1.1.7 | Edrophonium | 7-Acetoxy-4-methylcoumarin | Linear competitive inhibition KX = 0.14 μM | Berman (1990) |
| 12 | Acetylcholinesterase Torpedo californica | 3.1.1.7 | Phenyltrimethylammonium | 7-Acetoxy-4-methylcoumarin | Linear competitive inhibition KX = 17 μM | Berman (1990) |
| 13 | Acetylcholinesterase Torpedo californica | 3.1.1.7 | N-Methylacridinium | 7-Acetoxy-4-methylcoumarin | Linear competitive inhibition KX = 0.11 μM | Berman (1990) |
| 14 | Gamma-glutamyltransferase Homo sapiens | 2.3.2.2 | OU749 | Gly-Gly (10) | Competitive inhibition KX = 17.6 μM | King (2009) |
| 15 | Thrombin Homo sapiens | 3.4.21.5 | Recombinant hirudin | Tos-Gly-Pro-Arg-7-amino-4-methyl- coumarylamide | Competitive inhibition KX= 447 fM | Schmitz (1991) |
| 16 | Thrombin Homo sapiens | 3.4.21.5 | Hirudin1-47 fragment | Tos-Gly-Pro-Arg-7-amino-4-methyl- coumarylamide | Competitive inhibition KX= 0.42 μM | Schmitz (1991) |
| 17 | Thrombin Homo sapiens | 3.4.21.5 | Hirudin45-65 fragment | Fibrinogen Aα | Competitive inhibition KX= 0.76 μM | Schmitz (1991) |
| 18 | Thrombin Homo sapiens | 3.4.21.5 | Hirudin1-47 fragment | Fibrinogen Aα | Competitive inhibition KX= 0.46 μM | Schmitz (1991) |
| 19 | Protein-lysine desuccinylase (NAD+) Homo sapiens | 2.4.2.B14 | Compound 39 | Abz- GVLK(glutaryl)AY(NO2)GV-NH2 | Competitive inhibition KX= 83.2 nM | Kalbas (11) (2018) |
| 20 | Protein-lysine desuccinylase (NAD+) Homo sapiens | 2.4.2.B14 | Compound 39.2 | Abz- GVLK(glutaryl)AY(NO2)GV-NH2 | Competitive inhibition KX= 13.6 nM | Kalbas (11) (2018) |
| 21 | Coagulation factor Xa Homo sapiens | 3.4.21.6 | BAY 59-7939 (Rivaroxaban) | N-Methoxycarbonyl-D-cyclohexylalanyl- glycyl-L-arginine-4-nitroanilide acetate | Competitive inhibition KX= 0.4 nM | Perzborn (2005) |
| 22 | Acylglycerol lipase Homo sapiens | 3.1.1.23 | Compound 17b | 4-nitrophenylacetate | Competitive inhibition KX= 0.65 μM | Granchi (2016) |
| 23 | ABC-type quaternary amine transporter Mus musculus | 7.6.2.9 | Compound 9 | γ-Aminobutyric acid | Competitive inhibition | Al-Khawaja (2014) |
| 24 | beta-Galactosidase Escherichia coli | 3.2.1.23 | β-D-1-thiogalacto- pyranoside (IPTG) | Resorufin β-D-galactopyranoside | Competitive inhibition KX= 63 μM | Hess (12) (2015) |
| 25 | 3-Hydroxyacyl-[acyl-carrier-protein] dehydratase Yersinia pestis | 4.2.1.59 | Mangostin | Crotonyl-coenzyme-A | Competitive inhibition KX= 1.4 μM | McGillick (2016) |
| 26 | Caspase-3 Homo sapiens | 3.4.22.56 | Designed ankyrin repeat protein D3.4 | N-Acetyl-DEVD-7-amino-4-methylcoumarylamide | Purely competitive inhibition KX= 16.8 nM | Schroeder (2013) |
(1) Always the varied substrate. In two- or more-substrate reactions the concentration(s) of the non varied substrate(s) is/are kept constant.
(2) Name of the mechanism given by the authors in the quoted reference. α, β and the inhibition/activation constants for the modifier (X), uniformly denoted KX, are the values specified by the authors. In some cases, missing parameters have been calculated from graphical or tabular data provided in the papers. In two- or more-substrate reactions, KX represents an apparent constant at given concentrations of the fixed substrates and no calculations of the intrinsic values have been attempted.
(3) Full references at the end of the page provide also the digital object identifier (doi), if available. Clicking the authors (highlighted) opens the reference in PubMed.
(4) Heat desensitized enzyme, 24 h at 45°C.
(5) Approximate value calculated from the graphic in Fig. 5.
(6) A study dedicated to inhibition of spermine synthase (two substrates-two products reaction) by reaction products that suggests a compulsory-order mechanism in which both substrates bind the enzyme before release of products. See also the pages of LMx(Sp>Ca)I and LMx(Sp<Ca)I.
(7) In this study a truncated, His-tagged form of tyrosinase, which includes the catalytic domain, was used.
(8) This paper by Sun and coworkers is a recommended reading because it is a useful tutorial in the management of enzyme-inhibition kinetic data. The authors fit the general modifier mechanism model of Botts and Morales to an entire ensemble of data with a large number of experimental points. The analysis of inhibition mechanism is based on graphical representation of data and on statistical test that include the Akaike information criterion. See also results from this study under LMx(Sp>Ca)I and LMx(Sp<Ca)I.
(9) The study by Lorey et al. (2003) is centered on the inhibition of human dipeptidyl-peptidase IV by the peptide MDPVDPNIE, Tat(1-9), corresponding to the N-terminus of the human immunodeficiency virus-1 transactivator Tat (HIV-1 Tat, which consists of 86 amino acids), and derived peptides. See also cases of linear mixed, predominantly specific inhibition, with/without multiple inhibitor binding: LMx(Sp>Ca)I.
(10) Transpeptidation reaction of gamma-glutamyltransferase: (5-L-glutamyl)-peptide + amino acid = peptide + 5-L-glutamyl amino acid. See also LCaI.
(11) A synonym of Protein-lysine desuccinylase is Sirtuin 5. This is one of few papers in which the kinetic mechanism, LSpI, has been accurately demonstrated by showing the linear dependence of Kmapp on modifier concentration for twelve compounds analyzed in every due detail (paper and Supplement).
(12) The authors describe the innovative method Stroboscopic Epifluorescence Imaging for investigating enzyme kinetics, which allows ‘high time resolution and the ability to probe extremely large numbers of discrete reactions while consuming low sample volumes’. This must-read paper holds the record number of data points in a single experiment!
References
- Al-Khawaja A, Petersen JG, Damgaard M, Jensen MH, Vogensen SB, Lie MEK, Kragholm B, Bräuner-Osborne H, Clausen RP, Frølund B, Wellendorph P (2014) Pharmacological identification of a guanidine-containing β-alanine analogue with low micromolar potency and selectivity for the betaine/GABA transporter 1 (BGT1). Neurochem Res 39: 1988-1996. doi:10.1007/s11064-014-1336-9
- Barr JT, Jones JP (2011) Inhibition of human liver aldehyde oxidase: implications for potential drug-drug interactions. Drug Metab Disp 39: 2381-2386. doi:10.1124/dmd.111.041806
- Berman HA, Leonard K (1990) Ligand exclusion on acetylcholinesterase. Biochemistry 29: 10640-10649. doi:10.1021/bi00499a010
- Çokuğraş AN, Cengiz D, Tezcan EF (2004) Do alkylating agents modify the histidine residue of the desensitized butyrylcholinesterase? Protein J 23: 495-500. doi:10.1007/s10930-004-7876-0
- D’Adamio G, Sgambato A, Forcella M, Caccia S, Parmeggiani C, Casartelli M, Parenti P, Bini D, Cipolla L, Fusi P, Cardona F (2015) New synthesis and biological evaluation of uniflorine A derivatives: towards specific insect trehalase inhibitors. Org Biomol Chem 13: 886-892. doi:10.1039/C4OB02016B
- Granchi C, Rizzolio F, Palazzolo S, Carmignani S, Macchia M, Saccomanni G, Manera C, Martinelli A, Minutolo F, Tuccinardi T (2016) Structural optimization of 4-chlorobenzoylpiperidine derivatives for the development of potent, reversible, and selective monoacylglycerol lipase (MAGL) inhibitors. J Med Chem 59: 10299-10314. doi:10.1021/acs.jmedchem.6b01459
- Hess D, Rane A, deMello AJ, Stavrakis S (2015) High-throughput, quantitative enzyme kinetic analysis in microdroplets using stroboscopic epifluorescence imaging. Anal Chem 87: 4965-4972. doi:10.1021/acs.analchem.5b00766
- Kalbas D, Liebscher S, Nowak T, Meleshin M, Pannek M, Popp C, Alhalabi Z, Bordusa F, Sippl W, Steegborn C, Schutkowski M (2018) Potent and selective inhibitors of human sirtuin 5. J Med Chem 61: 2460-2471. doi:10.1021/acs.jmedchem.7b01648
- King JB, West MB, Cook PF, Hanigan MH (2009) A novel, species-specific class of uncompetitive inhibitors of gamma-glutamyl transpeptidase. J Biol Chem 284: 9059-9065. doi:10.1074/jbc.M809608200
- Lorey S, Stöckel-Maschek A, Faust J, Brandt W, Stiebitz B, Gorrell MD, Kähne T, Mrestani-Klaus C, Wrenger S, Reinhold D, Ansorge S, Neubert K (2003) Different modes of dipeptidyl peptidase IV (CD26) inhibition by oligopeptides derived from the N-terminus of HIV-1 Tat indicate at least two inhibitor binding sites. Eur J Biochem 270: 2147-2156. doi:10.1046/j.1432-1033.2003.03568.x
- McGillick BE, Kumaran D, Vieni C, Swaminathan S (2016) β-Hydroxyacyl-acyl carrier protein dehydratase (FabZ) from Francisella tularensis and Yersinia pestis: structure determination, enzymatic characterization, and cross-inhibition studies. Biochemistry 55: 1091-1099. doi:10.1021/acs.biochem.5b00832
- Ogawa T, Verhamme IM, Sun MF, Bock PE, Gailani D (2005) Exosite-mediated substrate recognition of factor IX by factor XIa. The factor XIa heavy chain is required for initial recognition of factor IX. J Biol Chem 280: 23523-23530. doi:10.1074/jbc.M500894200
- Pajula RL (1983) Kinetic properties of spermine synthase from bovine brain. Biochem J 215: 669-676. doi:10.1042/bj2150669
- Perzborn E, Strassburger J, Wilmen A, Pohlmann J, Roehrig S, K.H. S, Straub A (2005) In vitro and in vivo studies of the novel antithrombotic agent BAY 59‐7939—an oral, direct Factor Xa inhibitor. J Thromb Haemost 3: 514-521. doi:10.1111/j.1538-7836.2005.01166.x
- Schmitz T, Rothe M, Dodt J (1991) Mechanism of the inhibition of α-thrombin by hirudin-derived fragments hirudin(1-47) and hirudin(45-65). Eur J Biochem 195: 251-256. doi:10.1111/j.1432-1033.1991.tb15701.x
- Schroeder T, Barandun J, Fluetsch A, Briand C, Mittl PRE, Gruetter MG (2013) Specific inhibition of caspase-3 by a competitive DARPin: molecular mimicry between native and designed inhibitors. Structure 21: 277-289. doi:10.1016/j.str.2012.12.011
- Sun W, Wendt M, Klebe G, Röhm KH (2014) On the interpretation of tyrosinase inhibition kinetics. J Enzyme Inhib Med Chem 29: 92-99. doi:10.3109/14756366.2012.755621