Hyperbolic mixed, predominantly catalytic activation
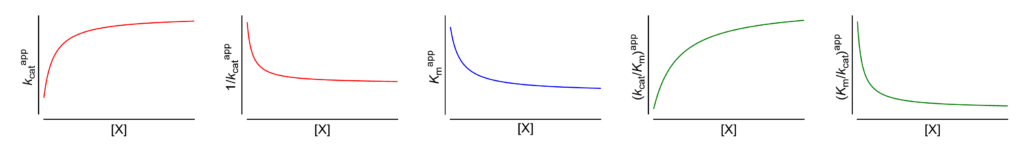
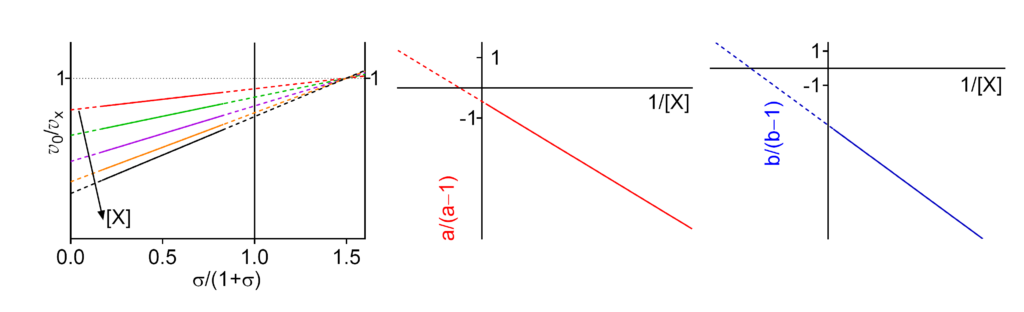
Featured examples
- Km↓ The apparent Michaelis constant decreases with increasing [X]
- V ↑ (∴kcat↑) The apparent limiting rate, and therefore the catalytic constant, increase with increasing [X]
These symbols are shown only when the featured dependencies of the parameters on modifier concentration have been demonstrated by the authors.
| # | Enzyme Species | EC no. | Modifier | Substrate(1) | Name given by authors (2) | Reference(3) |
|---|---|---|---|---|---|---|
| 1 | Thrombin Homo sapiens | 3.4.21.5 | Na+ | H-(D)-Phe-pipecolyl-Arg- p-nitroanilide | Activation α = 0.44, β = 2.38, KX = 0.02 M | Wells (1992) Di Cera (1995) |
| 2 | Thrombin Homo sapiens | 3.4.21.5 | hir55-65 (4) | H-(D)-Phe-pipecolyl-Arg- p-nitroanilide | Activation α = 0.34, β = 1.46, KX = 4.2 μM | Ayala (1994) Di Cera (1995) |
| 3 | Cathepsin K Homo sapiens | 3.4.22.38 | Dermatan sulfate | Cbz-Phe-Arg-7-amino-4-methylcoumarylamide | Nonessential activation α = 0.16, β = 1.25, KX = 9.2 μM | Novinec (2010) |
| 4 | 3-Phosphoshikimate 1-carboxyvinyltransferase Bacillus subtilis | 2.5.1.19 | NH4+ | Phosphoenol pyruvate (5) | Mixed-type nonessential K-type activation, Km↓ V-type activation, V↑ α ≈ 0.13, KX = 3.9 mM | Fischer (1987) |
| 5 | 3-Phosphoshikimate 1-carboxyvinyltransferase Bacillus subtilis | 2.5.1.19 | NH4+ | Shikimate-3-phosphate (5) | Mixed-type nonessential K-type activation, Km↓ V-type activation, V↑ α ≈ 0.15, KX = 3.9 mM | Fischer (1987) |
| 6 | Aldehyde dehydrogenase (NAD+) Homo sapiens | 1.2.1.3 | Alda-1 | NADox | Mixed-type nonessential activation, kcat↑, Km↓ α = 0.4, β = 1.6 | Belmont-Díaz (2016) |
| 7 | Cholesterol 24-hydroxylase (6) Homo sapiens | 1.14.14.25 | L-Glutamate | Cholesterol | Activation, kcat↑, Km↓ α = 0.56, β = 2.5 (7) | Mast (2017) |
| 8 | Cholesterol 24-hydroxylase (6) Homo sapiens | 1.14.14.25 | L-Glutamate | Cholesterol + NADPH-hemoprotein reductase (EC 1.6.2.4) as redox partner | Activation, kcat↑, Km↓ α = 0.4, β = 3.0 (7) | Mast (2017) |
| 9 | Pyruvate carboxylase Sus scrofa | 6.4.1.1 | Acetyl-CoA | MgATP2- | Activation α < 1, β > 1, KX ≈ 10-20 μM (8) | Warren (1974) |
| 10 | Tryptophan synthase Salmonella typhimurium | 4.2.1.20 | Na+ | Indole | Activation, V↑, Km↓ α = 0.03, β = 1.4, KX = 0.56 mM (9) | Peracchi (1995) |
| 11 | 1-Phosphatidylinositol phosphodiesterase Bacillus cereus | 3.1.4.10 | Butyl-fluorescein myo-inositol phosphate (activation by substrate) | Butyl-fluorescein myo-inositol phosphate | Activation α' = 0.2, β' = 1.4, Ks = 0.5 mM, K's = 0.15 mM (10) | Birrell (2003) |
| 12 | beta-Glucosidase Bacillus polymyxa | 3.2.1.21 | Glucose-6-phosphate | p-nitrophenyl β-D-glucopyranoside | Activation, α = 0.55, β = 1.83, KX = 25.0 μM | Weiss (2015) |
| 13 | Leukotriene-A4 hydrolase Homo sapiens | 3.3.2.6 | 4MDM | L-Ala-p-nitroanilide | Hyperbolic mixed, predominantly catalytic activation α = 0.82, β = 25.4, KX = 1.35 μM | Lee (2019) |
| 14 | Cholinesterase Homo sapiens | 3.1.1.8 | Tyramine | o-Nitrophenylacetate | Hyperbolic activation α < 1, β = 2.8 | Masson (2008) |
| 15 | Cholinesterase Homo sapiens | 3.1.1.8 | Tyramine | o-Nitroacetanilide | Hyperbolic activation α = 0.4, β = 5.5 | Masson (2008) |
(1) Always the varied substrate. In two- or more-substrate reactions the concentration(s) of the non varied substrate(s) is/are kept constant.
(2) Name of the mechanism given by the authors in the quoted reference. α, β and the inhibition/activation constants for the modifier (X), uniformly denoted KX, are the values specified by the authors. In some cases, missing parameters have been calculated from graphical or tabular data provided in the papers. In two- or more-substrate reactions, KX represents an apparent constant at given concentrations of the fixed substrates and no calculations of the intrinsic values have been attempted.
(3) Full references at the end of the page provide also the digital object identifier (doi), if available. Clicking the authors (highlighted) opens the reference in PubMed.
(4) C-terminal fragment of the natural inhibitor hirudin with the sequence Acetyl-DFEEIPEEYLQ-COOH.
(5) At saturating NH4+ concentration, both substrates (phosphoenol pyruvate and shikimate-3-phosphate) behave as nonessential activators according to the mechanism HMx(Sp>Ca)I (see under HMx(Sp>Ca)I).
(6) The truncated form Δ(2-50) of the enzyme was used to catalyze the reaction:
Cholesterol + [reduced NADPH-hemoprotein reductase] + O2 ⇄ (24S)-cholest-5-ene-3-beta,24-diol + [oxidized NADPH-hemoprotein reductase] + H2O
(7) Calculated from Fig. 5 in Mast (2017).
(8) Indicative values calculated from Fig. 2A in Warren (1974).
(9) Calculated from Table 1 in Peracchi (1995)
(10) Symbols as in the original paper [Birrell (2003), free full text, see References]. Enzyme with substrate alone, binding is cooperative. The enzyme is a small monomeric molecule with a catalytic site and an allosteric site that can bind either a second molecule of substrate, which triggers nonessential activation, or the activator dihexanoylphosphatidylcholine. When this is present, activation occurs in parallel through the substrate and the activator whose mechanisms is hyperbolic specific activation (see entry 5 in HSpA).
References
- Ayala Y, Di Cera E (1994) Molecular recognition by thrombin. Role of the slow to fast transition, site-specific ion binding energetics and thermodynamic mapping of structural components. J Mol Biol 235: 733-746. doi:10.1006/jmbi.1994.1024
- Belmont-Díaz JA, Yoval-Sánchez B, Calleja-Castañeda LF, Pardo Vázquez JP, Rodríguez-Zavala JS (2016) Alda-1 modulates the kinetic properties of mitochondrial aldehyde dehydrogenase (ALDH2). FEBS J 283: 3637-3650. doi:10.1111/febs.13833
- Birrell GB, Zaikova TO, Rukavishnikov AV, Keana JFW, Hayes Griffith O (2003) Allosteric interactions within subsites of a monomeric enzyme: Kinetics of fluorogenic substrates of PI-specific phospholipase C. Biophys J 84: 3264-3275. doi:10.1016/S0006-3495(03)70051-4
- Di Cera E, Dang QD, Ayala Y, Vindigni A (1995) Linkage at steady state: allosteric transitions of thrombin. Meth Enzymol 259: 127-144. doi:10.1016/0076-6879(95)59041-2
- Fischer RS, Rubin JL, Gaines CG, Jensen RA (1987) Glyphosate sensitivity of 5-enol-pyruvylshikimate-3-phosphate synthase from Bacillus subtilis depends upon state of activation induced by monovalent cations. Arch Biochem Biophys 256: 325-334. doi:10.1016/0003-9861(87)90453-X
- Lee KH, Petruncio G, Shim A, Burdick M, Zhang Z, Shim YM, Noble SM, Paige M (2019) Effect of modifier structure on the activation of leukotriene A4 hydrolase aminopeptidase activity. J Med Chem 62: 10605-10616. doi:10.1021/acs.jmedchem.9b00663
- Masson P, Froment MT, Gillon E, Nachon F, Lockridge O, Schopfer LM (2008) Kinetic analysis of effector modulation of butyrylcholinesterase-catalysed hydrolysis of acetanilides and homologous esters. FEBS J 275: 2617-2631. doi:10.1111/j.1742-4658.2008.06409.x
- Mast N, Anderson KW, Johnson KM, Phan TTN, Guengerich FP, Pikuleva IA (2017) In vitro cytochrome P450 46A1 (CYP46A1) activation by neuroactive compounds. J Biol Chem 292: 12934-12946. doi:10.1074/jbc.M117.794909
- Novinec M, Kovačič L, Lenarčič B, Baici A (2010) Conformational flexibility and allosteric regulation of cathepsin K. Biochem J 429: 379-389. doi:10.1042/BJ20100337
- Peracchi A, Mozzarelli A, Rossi GL (1995) Monovalent cations affect dynamic and functional properties of the tryptophan synthase a2b2 complex. Biochemistry 34: 9459-9465. doi:10.1021/bi00029a022
- Warren GB, Tipton KF (1974) The role of acetyl-CoA in the reaction pathway of pig-liver pyruvate carboxylase. Eur J Biochem 47: 549-554. doi:10.1111/j.1432-1033.1974.tb03724.x
- Weiss PHE, Álvares ACM, Gomes AA, Miletti LC, Skoronski E, da Silva GF, de Freitas SM, Magalhães MLB (2015) Beta glucosidase from Bacillus polymyxa is activated by glucose-6-phosphate. Arch Biochem Biophys 580: 50-56. doi:10.1016/j.abb.2015.06.012
- Wells CM, Di Cera E (1992) Thrombin is a Na+-activated enzyme. Biochemistry 31: 11721-11730. doi:10.1021/bi00162a008